Abstract Factors that determine the effectiveness of the digital pharmacovigilance database are considered in the example of the development and analysis of the OtiPharm® Data Pro platform. Factors were divided into two groups: highly specialized, in which compliance with legal requirements for pharmacovigilance has the highest-priority; and non-specialized, that influence on the user experience of […]


Quality, efficacy and safety are the factors that most concern the consumer when buying a medicines. Most people trust doctors in terms of effectiveness and quality of the medicines, but they tend to read about adverse reactions on the Internet, including social networks. According to the GlobalData survey for 2019, almost two thirds […]
Yesterday you took a new medicine and you suddenly had a stomach ache. After complaining of such trouble to the doctor, what should he do next? Side effect – an undesirable and dangerous effect of the medicines on the body, which occurs when using the medicines during treatment, diagnosis or prevention in the recommended doses. […]


Pharmacovigilance. How to simplify the work? Have you ever thought about the meaning of the word “simplify”? Is it about reducing the amount of work? We at OtiPharm Data Pro have long been looking for an answer to the question of how to make the work of a pharmacovigilance expert, QPPV, medical director […]
Pharmacovigilance is the activity of identifying, storing, evaluating, understanding, and preventing side effects or other potentially drug-related problems. During its operation, a large amount of paper and digital documentation are produced, which the pharmacovigilance manager (QPPV) needs to structure. Let’s look at a few of them. The first type: The simplest – storage […]
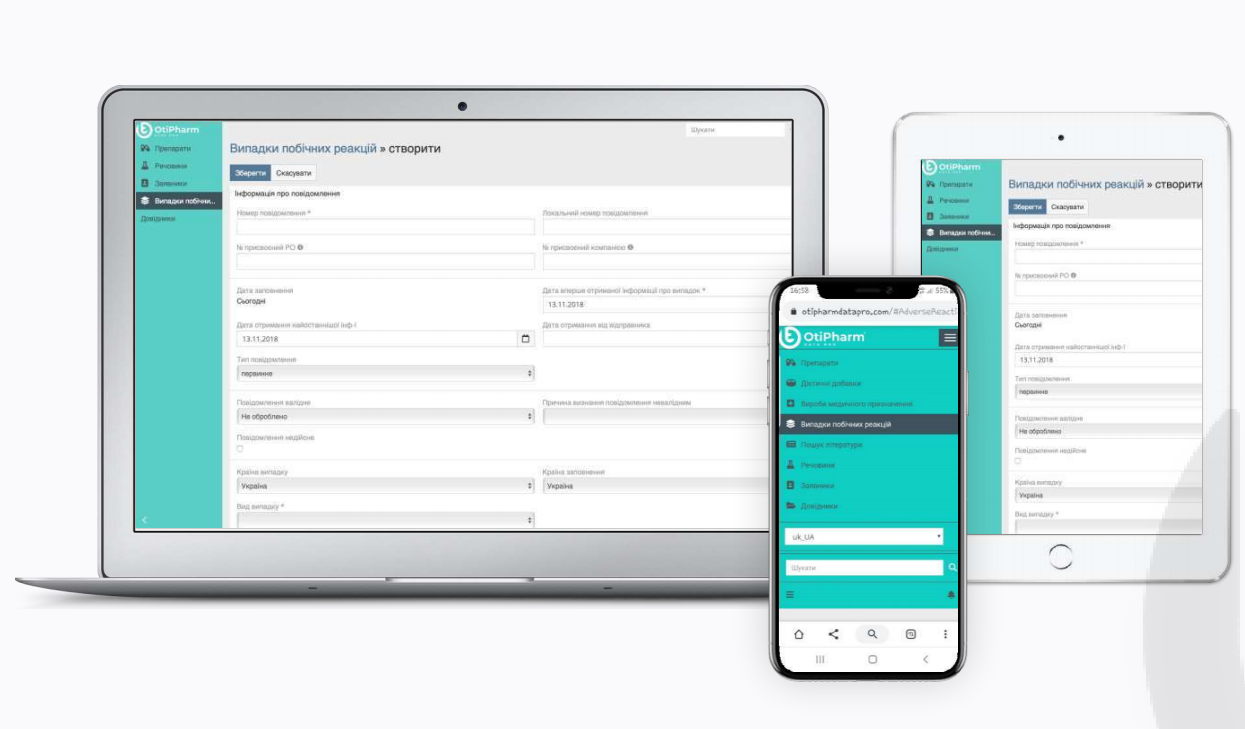

Pharmacovigilance as a working monitoring system has recently appeared in Ukraine, and immediately felt a crazy information load: Saving information about each medicines of the pharmaceutical company or the applicant, prompt changes; Obtaining and structuring data on adverse reactions and / or lack of efficacy from physicians, health care providers and patients; Systematic search and […]